All Type of News
Pharmacists, doctors differ over bill for prescription in question
Both pharmacists and doctors have mixed feelings over the passage of a bill that doctors should explain suspicious contents in prescription to a pharmacist who raise them.
The Korean Pharmaceutical Association said...
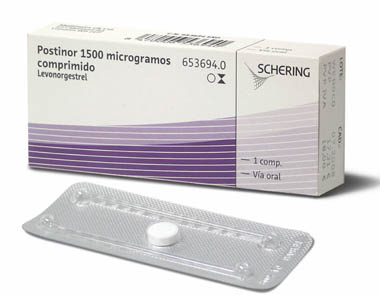
Bayer Healthcare to launch an emergent contraceptive pill.Bayer Healthcare (Bayer Schering Pharma) has launched Postino-1, an emergent oral contraceptive pill to add a full pipeline of contraceptive products including an oral pill, IUD...
|
Sanofi-Aventis Korea launches 24-hour blood pressure measurement campaign
Sanofi-Aventis Korea has unveiled its new campaign to raise awareness about hypertension (high blood pressure), a leading cause of heart disease and stroke.
As part of its efforts to encourage blood pressure manage...
KFDA won legal battle on bioequivalency manipulation case.
The KFDA won the lawsuit against the case of drug manipulated bio equivalency tests earlier raised by Schnell Pharm. Korea. The Seoul Administrative court decided to reject the lawsuit raised by Schnell Korea on the s...
EDI reported Plavix as the top prescription drug in 2006.
Plavix, a foreign brand prescription drug of Sanofi-Aventis, showed the top on the EDI bill changes last year while Amodipine of Hanmi kept the top among the local brands according to the 2006 EDI bill report.
...
Concerns are rising over less benefits in minor disease
The government's plan to lower health insurance benefits for treatment of minor diseases, such as common cold, from August has sparked a concern that the plan will significantly reduce the number of prescriptions, des...
Lilly to launch anti-osteoporosis drug, Fosteo.
Lilly Korea announced to launch Fosteo (teriparatide), a unique anti-osteoporosisdrug, for the promotion of bone formation.
Fosteo is a parathyroidal hormone to promote a bone formation instead of blocking an excre...
Use of RFID tags to be mandatory in drugs
The government plans to employ the radio frequency identification (RFID) or smart sensor technologies in drugs and foods, starting to replace the current barcodes as early as next year.
The RFID tag is a small inte...
Exelon patch to be available in Korea
Exelon patch, the first transdermal therapy for Alzheimer's disease, will be likely approved by the Korea Food and Drug Administration, possibly within this year, based on the results of the IDEAL (Investigation of Tr...
CTD system for new drugs to be made compulsory in 2009
Drug makers will be obliged to make their application for new drugs based on the common technical document (CTD) guidelines from March 2009, according to the Korea Food and Drug Administration.
The administration a...